- More than 250 potential COVID-19 vaccines are being developed currently worldwide.
- 10 billion – the estimated number of vaccine doses that would need to be delivered to all corners of the world within two years.
When the coronavirus became a global pandemic in early 2020, the life sciences and healthcare (LSH) supply chain faced unprecedented challenges. Although DHL quickly put crisis management plans into action, our teams still had to remain alert and agile in order to respond to a rapidly changing environment. There was little time to plan ahead.
More recently we’ve been able to look back on lessons learned and focus our attention on the future. And none too soon, because the next hurdle is already visible on the horizon: transporting, warehousing and distributing COVID-19 vaccines around the world.
Right now, more than 250 potential COVID-19 vaccines are in various stages of development. It’s possible that one could be ready for distribution by the end of 2020. Because of the urgency of the pandemic and the sheer volume of potential doses on the way, DHL is preparing to secure stable supply chains for vaccines and medical supplies for this crisis and beyond. Although many questions remain unanswered, what is clear is that we need to plan ahead and understand the potential range of requirements and their implications for logistics.
Will the cold chain need to get colder?
Today’s conventional cold supply chains transport temperature-sensitive medical supplies at temperatures between two and eight degrees Celsius. This includes vaccines, which must be stored properly from the time they are manufactured until they are administered. Any exposure to inappropriate conditions, such as higher temperatures, could reduce or even destroy the vaccine’s potency.

Normally researchers devote tons of time to testing vaccine stability in order to understand the environmental conditions under which the vaccine can be securely transported or stored. They use the data generated to improve stability and make doses easier to distribute. The current COVID-19 vaccines are being developed at such unprecedented speeds that researchers are fully focused on safety and efficacy in order to get them approved as quickly as possible.
The lack of stability data may lead some producers, out of caution, to require extreme temperatures along the supply chain (as low as -80 °C). We’ve already seen similar requirements in certain COVID-19 vaccine clinical trials.
Fully understanding the range of potential temperature requirements and their implication for logistics allows us to map out the supply chain, identify potential bottlenecks and plan end-to-end solutions.
What will an end-to-end solution look like?
At DHL, we expect a number of challenges along the supply chain. On the production end, the logistics infrastructure is largely intact, making vaccine shipments more manageable. However, highly fluctuating processes, such as quality assurance and customs clearance, can make maintaining cooling requirements more difficult.
Given the urgency of the pandemic, longer distances will need to be covered by air. Here the sheer volume of shipments will be a challenge. Assuming 10 billion doses are to be distributed over two years, DHL estimates the need for 15 million deliveries in cooling boxes, 200,000 movements by pallet shippers and 15,000 flights across the various supply chain setups.
Downstream it will become increasingly difficult to comply with stringent temperature requirements, especially in certain parts of the world. Currently, large parts of Africa, South America and Asia could not be readily supplied at scale due to lack of cold-chain logistics capacity suitable for life science products. It’s going to take extraordinary measures to reach people in these places, where some two-thirds of the world’s population live. Governments and NGOs need to start improving infrastructure now and collaborating closely with vaccine producers and logistics providers to ensure doses can be distributed to everyone as soon as vaccines are available.
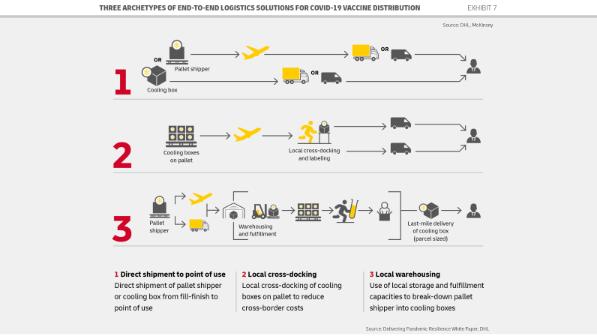
Logistics providers also need to be ready with multiple end-to-end solutions to meet a variety of needs. At DHL, we’ve identified three supply chain archetypes – direct shipment to point of use, local cross-docking, and local warehousing. The suitability of each depends on the temperature requirements, transport distances, and volumes as well as factors related to cost, lead time, warehouse capacity and the availability of packaging and equipment.
Why go looking for the latest logistics stories and insights when you can have them delivered right to you?
Delivering Pandemic Resilience
DHL’s latest white paper Delivering Pandemic Resilience dives much deeper into these issues. In the study, we draw on our decades of global logistics and supply chain experience, as well as analytical support from McKinsey & Company, to identify critical pain points along the COVID-19 supply chain, outline the potential requirements and possible solutions for distributing a COVID-19 vaccine around the world, and offer a five-pillar framework to tackle future emergencies.
I encourage you to review and share it with your colleagues and partners. After all, the coronavirus pandemic will not be the last global health crisis. It’s time for governments, NGOs, vaccine producers and logistics providers to work together, plan ahead, and be prepared for the challenge of COVID-19 vaccine logistics and beyond.
Published: October 2020







